Published on Mar 02, 2021
Last modified on May 05, 2025
Elektra Guide To Hormone Replacement Therapy (HRT)
5 min read
One of the most hotly debated and misunderstood topics surrounding menopause is…drum roll, please…hormone replacement therapy, or HRT for short. Many books (textbooks, even) have been written on the subject, but fortunately our team has done the legwork for you and distilled all the must-know info into an easy-to-understand guide. There’s a lot of doctor-reviewed information here so if you have any questions, book an appointment with an Elektra provider — we’re here for you!
What is HRT?
During the menopausal transition, the body’s production of two key female hormones, estrogen and progesterone, begins to fluctuate and progressively decline. HRT involves taking a supplemental form of either estrogen, or estrogen and progesterone together, to balance out and replace what the body is no longer producing. Additional hormones such as testosterone and DHEA are sometimes used as well, but for the most part, HRT refers to estrogen only or estrogen and progesterone combos.
Here’s a refresher on what both hormones do and why they’re important:

What’s in a name?
HRT goes by many names, including hormone therapy (HT), menopausal hormone therapy (MHT), and estrogen replacement therapy (ET). They’re all different ways of referring to medication containing only estrogen, or estrogen and progesterone together.
When to use HRT
HRT is most often prescribed to women with moderate to severe symptoms of menopause. In addition, it may be recommended to women who experience premature or early menopause (when periods stop before age 40) and those at significant risk for bone loss or osteoporosis HRT is also commonly prescribed to women who undergo a surgical procedure called an oophorectomy (truly, a tongue-twister). This is when one or both ovaries are removed, and sometimes takes place at the same time as a hysterectomy, when the uterus is removed.
HRT is one of several powerful tools in our toolbox of menopause treatments.
It can deliver short-term and long-term health benefits for women who are appropriate candidates for it. The North American Menopause Society, the American Society for Reproductive Medicine, and The Endocrine Society take the position that most healthy, recently menopausal women can use hormone therapy for relief of their symptoms of hot flashes and vaginal dryness if they and their providers determine that the benefits outweigh the risks based on their personal history. Read on to learn more about these benefits and risks, and the nuances around the history of HRT that have dampened its reputation.

HRT’s controversial history
If you have heard HRT causes cancer, you’re not alone. Let’s take a step back for a sec to unpack this history, shall we?
Hormone therapy was once routinely used to treat menopausal symptoms and protect long-term health — so much so that by 1992, Premarin (an estrogen pill) was the best-selling drug in the US, with ~40% of women aged 50+ taking HRT.
And then 2002 rolled around, when prematurely published findings of a massive study — The Women’s Health Initiative (WHI) — suggested that HRT usage led to a higher risk of breast cancer, heart disease, and blood clots. This set off a media *uproar* that led many women and doctors to abandon HRT practically overnight — we’re talkin’ 80% quitting cold turkey.
What many failed to notice, however, was that the sample group used in the initial WHI findings were already in a high risk category in terms of age and health. In fact, a significant group of WHI subjects saw improved (yes, improved) health outcomes on HRT, including decreased heart disease and breast cancer.
Soooo…is it safe?
Since the WHI study was published in 2002, hundreds of follow-up studies and clinical trials have been conducted in the United States by the National Institutes of Health (NIH) and other academic institutions. Today, the North American Menopause Society (NAMS) considers HRT to be safe and effective for most women, in perimenopause – menopause, especially when started prior to age 60 or within 10 years of menopause.
“Further analysis of the WHI data has shown hormone therapy to be safe and effective for menopausal women who have bothersome hot flashes, night sweats, or sleep disruption if they start hormone therapy while aged younger than 60 or within 10 years of menopause,” confirmed NAMS Executive Director Dr. JoAnn Pinkerton. “Unfortunately, fear continues to keep symptomatic women from being offered, or using, hormone therapy.”
Hormone therapy is safe and effective for menopausal women if they start hormone therapy while aged younger than 60 or within 10 years of menopause.
As with all medications, there are risks that have to be weighed in the context of an individual’s needs, goals, and health history. Furthermore, HRT is not a good option for those with a history of blood clots, cardiac or liver disease, or breast cancer.
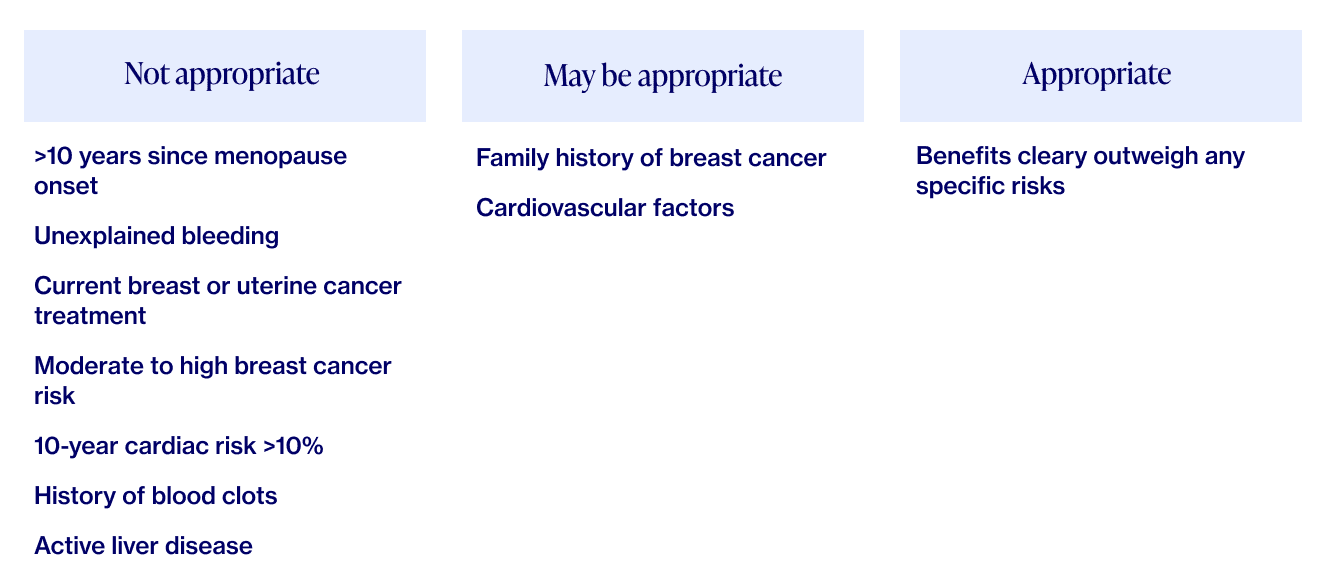
Menopause specialists, including those at Elektra Health, are trained to use two validated tools as a way to evaluate individual risk factors via algorithms for cardiovascular disease and breast cancer. These tools, along with your preferences, health status and your family’s health history, can help your provider determine whether HRT will be a good fit for you, and to tailor your treatment accordingly.
Curious and want to learn more about the benefits and side effects of HRT? Ask your existing healthcare provider or Elektra — we’re here to provide answers.
HRT and Breast Cancer
Despite the updated stance on HRT safety, many women, when they hear HRT, still immediately think…but wait, doesn’t that cause breast cancer?
Several recent studies have undermined the connection between HRT and breast cancer. One of the most prominent and widely publicized was the WHI follow-up study, the results of which were presented at the San Antonio Breast Cancer symposium in 2019. The primary finding?
Postmenopausal estrogen-only therapy is associated with a lower risk of breast cancer.
To be clear, this applies to estrogen only, not estrogen and progesterone. To put this in perspective: the absolute risk for breast cancer is 24/1000 women over 10 years. For women taking estrogen alone, there were 5 LESS breast cancer cases, but for women taking estrogen + progesterone, there were 6 MORE cases.
Although we still don’t have scientific explanations as to why estrogen alone produces these results, it’s promising nonetheless and a testament to the fact that this specific type of HRT is much safer than we originally thought when it comes to breast cancer risk and, as a result, a more viable option for menopausal symptom relief.
Types of HRT
There are over 10 types of HRT, which can be broken down into categories based on origin, composition, type of manufacturing, and whether they affect the whole body or are active locally.
Delivery type
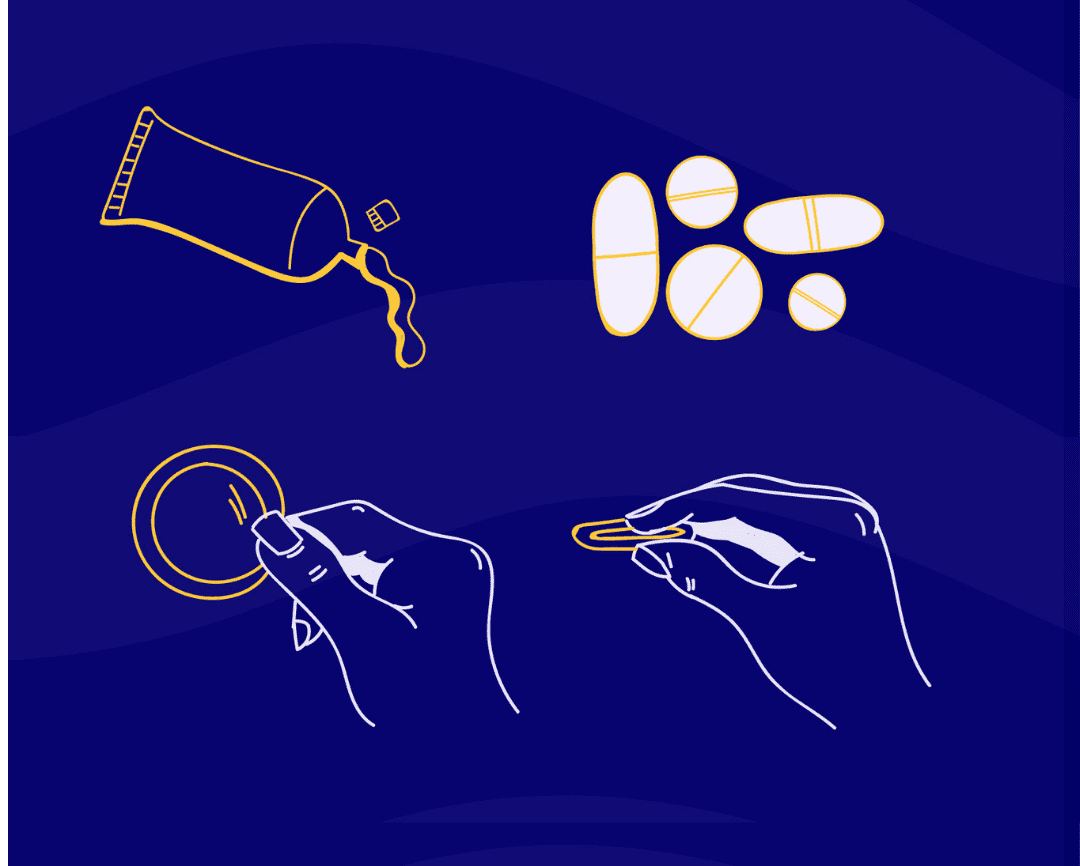
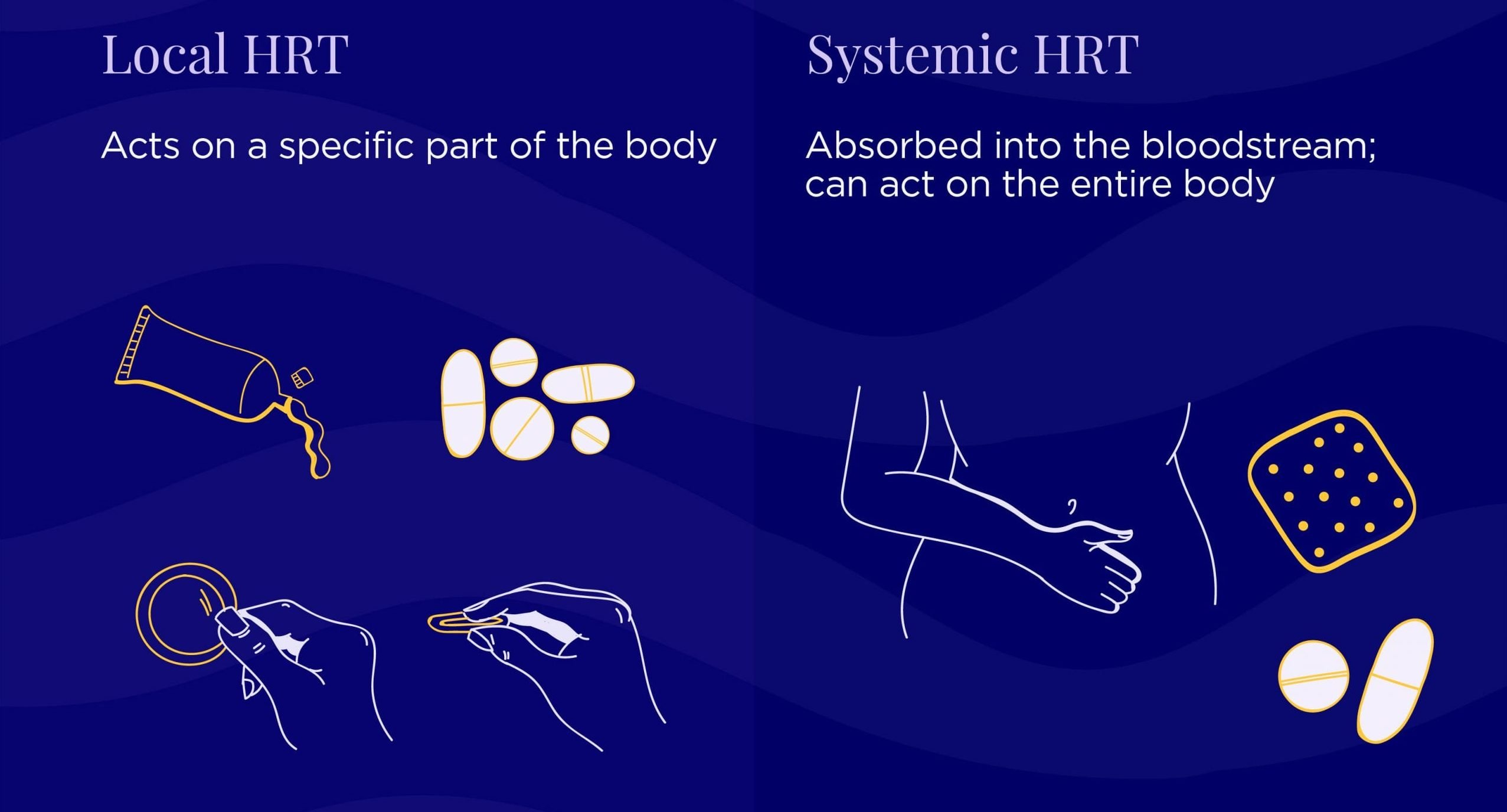
Local: acts on a specific part of the body for localized results (e.g. vaginal dryness & urinary symptoms)
Examples: vaginal estrogen tablets, creams, rings, vaginal DHEA suppositories (a hormone produced naturally in your adrenal glands that is converted by the body into estrogen)
Systemic: absorbed throughout the body for systemic results (e.g. hot flashes, night sweats, PMS)
Examples: estrogen (or estrogen & progesterone) gels, patches, pills, creams, etc.
Origin / composition type
Bioidentical: when the structure of the product’s active ingredient has the same chemical & molecular structure as the human body
Note that although bioidentical hormones are considered “natural,” they are still processed in a lab.
Synthetic / Non-bioidentical: when the product’s active ingredients are chemically synthesized, altered in a lab, or come from a non-human source such as Premarin (pregnant horses’ urine – yes, that is correct)
Manufacturing type
Compounded: Custom-made at a special pharmacy
FDA-approved: Standardized, consistent & studied for safety by the Food and Drug Administration
Note that many women do not realize that regular FDA-approved hormone prescriptions can be written for “bioidentical” hormones and do not need to be compounded.

Elektra does not recommend compounded hormones as a first line of treatment for the following reasons:
- They’re not regulated by the FDA and their claims around fewer risks/side effects & increased efficacy cannot be validated
- They may be associated with a slightly higher risk of endometrial cancer (in a study conducted by NAMS, there were 4 cases of endometrial cancer in the group using compounded hormone therapy compared to zero cases in the group using commercial products)
Instead, we recommend FDA-approved bioidentical hormones which offer greater safety, purity, and reliability — and, compared to synthetic hormones, decreased risk of breast cancer.
That’s not to say compounded hormones have NO place in the treatment of menopause. However, they may be prescribed in the case of an allergy or special circumstances that require looking beyond FDA-approved medications.
Forms of HRT
Oral
As the name suggests, oral products are taken by mouth in tablet or capsule form. Some contain a single hormone (estrogen) while others carry a combination of estrogen and progesterone or progestin (the synthetic form of the body’s naturally-occurring progesterone). Estrogen-only therapy is used for women who have had a hysterectomy, while combination therapy is for women with a uterus.
Oral treatments may be either sequential or combined:
Sequential HRT mimics your natural cycle. Estrogen is continuous but progesterone is sequential and only released for part of the month. Combined, on the other hand, is when estrogen and progesterone are taken together for every day of your cycle.
Transdermal
Transdermal products are topical and applied to the skin via creams, patches, sprays, or gels that are absorbed directly into the bloodstream. They’re applied either once daily, twice weekly, or weekly to the arm, shoulder, legs, or abdomen.
Vaginal
Low-dose estrogen can be applied directly to the vagina in the form of creams, a vaginal ring (which needs to be replaced every three months), or an estrogen tablet. This method is tailored towards women who are experiencing vaginal dryness, itchiness, and/or burning pain during intercourse. Examples include Vagifem and IMVEXXY.
Because low-dose vaginal estrogen (such as Vagifem) has not been found to affect systemic estrogen levels in clinical studies, it can be a viable choice for women with a high breast cancer risk who are seeking a solution for vaginal dryness.
HRT vs birth control…what’s the difference?
Some doctors advise perimenopausal women to take low-dose birth control pills as a way to regulate cycles. Compared to typical birth control pills, which contain 10-50 micrograms of estrogen, estrogen prescribed to women for menopause at the lowest dose contain only 10-20 micrograms. In other words, estrogen prescribed for menopause can be a smaller fraction (⅓ to ½ ) of the estrogen in a birth control pill. In addition to cycle regulation, the benefits of low-dose birth control include protection from ovarian/uterine cancer and the prevention of bone loss.
The key difference between low-dose birth control and HRT is that, unlike birth control, HRT doesn’t suppress the production of your own hormones. Instead, it adds hormones to your body to balance out what you’re not producing enough of. That’s why HRT is most often prescribed just as you reach the postmenopause threshold.
So, what’s the bottom line on HRT?
HRT has been found to be safe for most women experiencing menopause-related symptoms but, as with all medications, there are also potential downsides. Here are the current facts: Family history and specific circumstances can play a major role here, so it’s extremely important to take a personalized and individualized approach to decisions about HRT. That approach is embedded in Elektra’s philosophy of care because that’s how we ourselves wish to be cared for.
It’s a lot to take in so we’ll summarize briefly:
- HRT balances out fluctuating and declining hormones to relieve menopause symptoms, and provides hormonal stability when the levels are very low
- HRT is safe for most women, but risks vs. benefits should be assessed individually for each person – this approach is at the heart of Elektra’s philosophy of care
- HRT can be used locally for vaginal symptoms or systemically for full-body symptoms like hot flashes through a variety of means – from pills to patches to gels

HRT should always be a discussion between you and a provider who is specifically trained in menopause. Together, you should review the risks and benefits of HRT and come to a decision that fits your goals and needs.
Don’t have a provider trained in menopause care? Contact Elektra ([email protected]) or book an appointment with one of our women’s health experts.
Alternatives to HRT
While HRT might be a safe and effective treatment for many, it’s not for everyone. If your family medical history or underlying medical condition precludes you from opting for this treatment, not to worry. The same is true if you’re not comfortable with exploring HRT for any other reason.
There are several safe and effective non-hormonal medications and supplements that can be used to treat menopause symptoms and improve quality of life. As with HRT, these are best discussed with your healthcare provider. And of course, lifestyle can play a huge role in symptom management, too.
For further reading
“Hormone therapy, long shunned for a possible breast cancer risk, is now seen as a short-term treatment for menopause symptoms” (Washington Post)
“Taking Hormones for Menopause Doesn’t Raise Early Death Risk” (The New York Times)
Hormone Therapy & Menopause FAQs (The North American Menopause Society)
Reviewed by Dr. Anna Barbieri, MD – last reviewed 11/4/21
Disclaimer: This information is for general educational purposes, and should not be used as a substitute for medical advice, diagnosis, or treatment of any health condition or problem.
As with anything you put into your body, taking dietary supplements can also involve health risks. You should consult a medical professional before taking supplements and inform your doctor about any supplements, as well as any medications you already take, since there may be interactions.
References
Hormone Replacement Therapy for Menopause. (2002, October 7). WebMD.
Frequently Asked Questions About Menopause. (2002, October 7). WebMD.
HRT vs Birth Control Pills: The Ultimate Guide. (n.d.). EarlyMenopause.Com. Retrieved December 7, 2020.
Hormone Therapy Products Chart. (2020, September 26). Women Living Better.
Manson, J. A. E., Chlebowski, R. T., Stefanick, M. L., Aragaki, A. K., Rossouw, J. E., Prentice, R. L., Anderson, G., Howard, B. V., Thomson, C. A., LaCroix, A. Z., Wactawski-Wende, J., Jackson, R. D., Limacher, M., Margolis, K. L., Wassertheil-Smoller, S., Beresford, S. A., Cauley, J. A., Eaton, C. B., Gass, M., … Wallace, R. B. (2013). Menopausal Hormone Therapy and Health Outcomes During the Intervention and Extended Poststopping Phases of the Women’s Health Initiative Randomized Trials. JAMA, 310(13), 1353.
Landau, M. D., & Church, K. (2018, August 20). What Is the History of Hormone Therapy, or HRT? | Everyday Health. EverydayHealth.Com.
Wolff, B. J. (2018). A Brief History of Treating Menopause. AARP.
Buist, D. S. M., Newton, K. M., Miglioretti, D. L., Beverly, K., Connelly, M. T., Andrade, S., Hartsfield, C. L., Wei, F., Chan, K. A., & Kessler, L. (2004). Hormone Therapy Prescribing Patterns in the United States. Obstetrics & Gynecology, 104(5, Part 1), 1042–1050.
Clark. (2007). A critique of Women’s Health Initiative Studies (2002-2006). Nuclear Receptor Signaling, 4. Link.
The 2017 hormone therapy position statement of The North American Menopause Society. (2017). Menopause, 24(7), 728–753.
Lobo, R. A. (2013). Where Are We 10 Years After the Women’s Health Initiative? The Journal of Clinical Endocrinology & Metabolism, 98(5), 1771–1780.
Ragaz, J., Qian, H., Wong, H., Wilson, K. S., Shakeraneh, S., & Spinelli, J. J. (2019). Abstract P6-13-04: Estrogen-alone based hormone replacement therapy (HRT) reduces breast cancer (BrCa) incidence and mortality whereas estrogen plus progestin Provera based HRT increases both BrCa incidence and BrCa mortality: A comparative analysis of Women’s Health Initiative trials. Poster Session Abstracts.
Melville, N. A. (2020, July 26). Younger Women Misinterpret Risks of HRT, Forgo Benefits. Medscape.
Saling, J. (2012, October 2). DHEA Supplements. WebMD.
Gass, M. L. S., Stuenkel, C. A., Utian, W. H., LaCroix, A., Liu, J. H., & Shifren, J. L. (2015). Use of compounded hormone therapy in the United States. Menopause, 22(12), 1276–1285.
Streicher, L. (2019, November 21). Buyer Beware: “Bioidentical” Hormone Myths. Dr. Lauren Streicher.
Files, J. A., Ko, M. G., & Pruthi, S. (2011). Bioidentical Hormone Therapy. Mayo Clinic Proceedings, 86(7), 673–680.
M. (2018, January 24). Hormone Replacement Therapy (HRT) part 3 ~. Hormone Health.
The Use of Vaginal Estrogen in Women With a History of Estrogen-Dependent Breast Cancer. (n.d.). ACOG.
The North American Menopause Society. (2017, July). Scientific Background Report for the 2017 Hormone Therapy Position Statement of The North American Menopause Society. Menopause.
Pinkerton, J. A. V. (2020). Hormone Therapy for Postmenopausal Women. New England Journal of Medicine, 382(5), 446–455.
MacLennan, A. H., Broadbent, J. L., Lester, S., & Moore, V. (2004). Oral oestrogen and combined oestrogen/progestogen therapy versus placebo for hot flushes. Cochrane Database of Systematic Reviews.
Management of Menopausal Symptoms. (2014, January). ACOG.
Manson, J. A. E., Aragaki, A. K., Rossouw, J. E., Anderson, G. L., Prentice, R. L., LaCroix, A. Z., Chlebowski, R. T., Howard, B. V., Thomson, C. A., Margolis, K. L., Lewis, C. E., Stefanick, M. L., Jackson, R. D., Johnson, K. C., Martin, L. W., Shumaker, S. A., Espeland, M. A., & Wactawski-Wende, J. (2017). Menopausal Hormone Therapy and Long-term All-Cause and Cause-Specific Mortality. JAMA, 318(10), 927.
Manson, J. A. E., Ames, J. M., Shapiro, M., Gass, M. L. S., Shifren, J. L., Stuenkel, C. A., Pinkerton, J. A. V., Kaunitz, A. M., Pace, D. T., Kagan, R., Schnatz, P. F., Kingsberg, S. A., Liu, J. H., Joffe, H., Richard-Davis, G., Goldstein, S. R., Schiff, I., & Utian, W. H. (2015). Algorithm and mobile app for menopausal symptom management and hormonal/non-hormonal therapy decision making. Menopause, 22(3), 247–253.
Merck Manuals. (2019, December). Menopause. Merck Manuals Professional Edition.
Type and timing of menopausal hormone therapy and breast cancer risk: individual participant meta-analysis of the worldwide epidemiological evidence. (2019). The Lancet, 394(10204), 1159–1168.
Experts Agree About Hormone Therapy, Menopause Relief | The North American Menopause Society, NAMS. (n.d.). The North American Menopause Society.


